Increasing the number of piglets weaned from hyper-prolific sows
Published Tuesday, 29th May 2018
Live yeasts have the potential to reduce lightweight pigs at birth and increase the number of piglet weaned through a multi-mechanistic action, says AB Vista’s Dr Pete Wilcock.
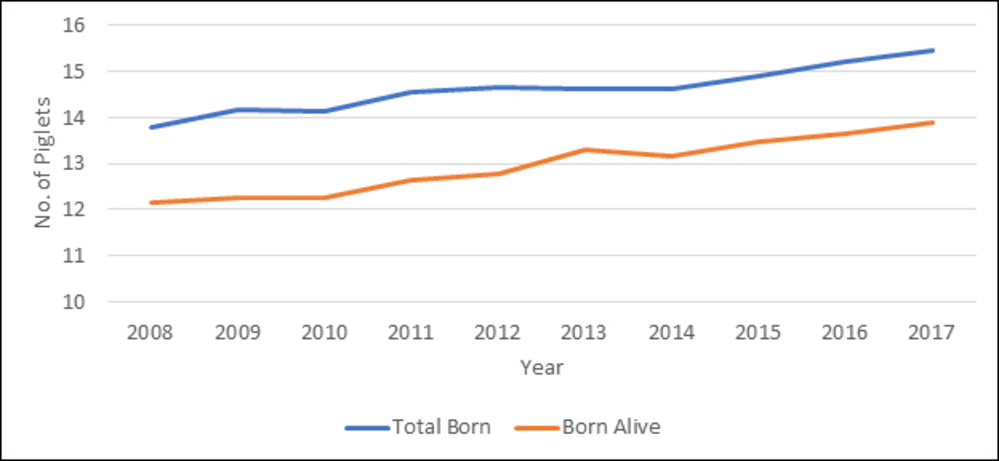
With improving genetics, the hyper-prolific sows of today continue to increase the number of piglets born per litter. Over the last 10 years there has been an average yearly increase in piglets born of 0.22 per year for the mean, and 0.16 pigs (Figure 1) for the top 10% performing producers in the USA (adapted from PigCHAMP Benchmarking, 2017).
Figure 1. The average number of piglets born and born alive by year for the top 10% of producers recorded in the US (PigChamp Benchmarking Summaries; 2008-2017)
As litter size increases, this brings other well recognized challenges to the production system; lower average birth weights and, more importantly, a higher percent of piglets in the lower birthweight category of < 1 kg. Often these lighter pigs will have lighter weaning weights, higher rates of pre-weaning mortality and take more days to market.
As an example, a European research group (Diaz et al., 2016) looked at the impact that birthweight (1050 pigs) can have on subsequent pig performance and concluded that a 100 g difference at birthweight could translate into a 1.3 kg difference in carcass weight. In addition, this study and a US study (Bergstrom et. al, 2009) both showed that pigs that were < 1.05 kg at birth had an overall higher mortality during lactation; 30% and 24% respectively.
Tools, both management and nutritional, that can help mitigate these effects become important to ensure that the higher production efficiency of the hyper-prolific sows is optimized.
Yeast products
One such area of nutritional research that has gained more interest with the increased level of sow productivity is the use of yeasts. There are a number of yeasts and yeast derivatives used in the animal feed industry, including live yeasts, yeast culture, yeast cell wall, yeast β-glucans, yeast α-mannans, and yeast extracts including peptides and nucleotides.
Live yeasts are mostly active dry yeasts (typically Saccharomyces cerevisiae) and in the US are defined as having a minimum content of 15 billion viable colony forming units (CFU) per gram, with levels typically ranging between 15-20 billion CFU/g, depending on the strain.
In contrast, yeast culture is produced from a culture media inoculated with yeast to allow fermentation, and then dried so no or limited viable yeast cells remain. This fermentation process results in metabolites that can be beneficial to the animal.
Yeast components
Yeast cell walls are typically composed of carbohydrates found in the cell wall and consist predominantly of glucans and mannans. β-glucans are the main polysaccharide component of the yeast and are composed mainly of β (1-3) linked glucose molecules with β (1-6) sidechains. It is well documented that yeast β-glucan can act as an immune-modulator through its ability to bind to specific carbohydrate receptors that are found on, for example, macrophages and antigen presenting cells. This stimulates a series of pathways that leads to the enhancement of the innate immune system when undergoing a challenge.
A study (Li et al., 2014) in piglets showed that when fed a 50 mg/kg dose of β-glucans, anti-inflammatory cytokines such as IL-10 were increased, while the pro-inflammatory cytokines such as TNF-alpha and IL-6 were reduced. This relationship between pro-inflammatory and anti-inflammatory cytokines plays an important role in the animal’s immune response and a reduction in pro-inflammatory cytokines may allow pigs to achieve a greater level of performance.
Mannans are linked in a branch structure primarily by α (1-6) bonds, with mannose linked to the backbone by α (1-2) bonds. The mannans can play an important role in reducing the pathogenic challenge to an animal as they can bind to certain E. coli and Salmonella, preventing them from binding to the gastro-intestinal tract and, in effect, flushing the pathogens out of the system, thereby limiting their ability to colonize. Yeast cell wall and live yeast have similar capabilities to bind both E. coli and Salmonella (Walker et al., 2013, Posadas et al., 2017).
Nucleotides are also found in relative high concentrations of up to 10% within whole yeast, and it is recognized that they can play a role in intestinal integrity, functionality, and the immune response.
Reducing lightweight pigs at birth
Although the use of live yeasts in ruminants is well defined, it is less understood in swine but it is likely a multi-mechanistic effect. Live yeast is a probiotic and acts as an oxygen scavenger, whereby it can potentially modify the environment of the large intestine of the pig, making it more anaerobic.
This is potentially favorable to fiber-degrading bacteria, and can result in increased levels of NDF breakdown and volatile fatty acids (Lizardo et al., 2012), and thus in a higher level of energy utilization. Coupled with the immunomodulation characteristics and the ability to limit pathogenic binding, the use of live yeast in sows to support piglet productivity presents an opportunity for the producer to optimize the production efficiency of the hyper-prolific sow.
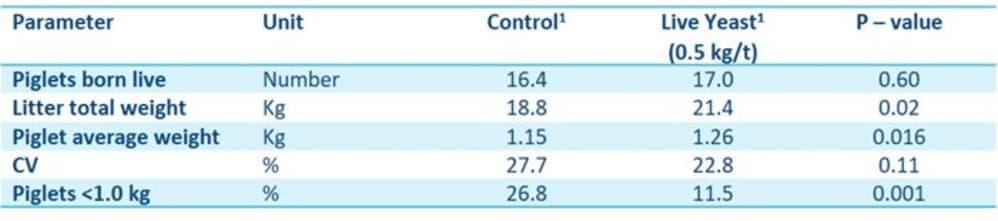
A recent study by AB Vista examined the effect that live yeast (20 billion CFU/g), when dosed at 0.5 kg/t during gestation, can have on sow farrowing performance. Although there was no effect on total piglets born between treatments, piglets from sows fed diets with live yeast showed a higher litter and piglet birth weight and, more critically, a lower percentage of piglets < 1 kg (Table 1). This shows the potential role that live yeast may play in mitigating the number of small pigs in the system, and justifies an evaluation in larger scale commercial systems.
Table 1. Farrowing performance of sows fed a control diet or that diet supplemented with 0.5 kg/t live yeast
Increasing the number of piglets weaned
The addition of a live yeast in gestation and lactation can also boost the immunoglobulin (Ig) content of colostrum and milk (Zanello et al., 2013). Improving the Ig content of colostrum and milk is advantageous as this provides direct immune protection to the naive piglet sucking on the sow. Of particular interest is IgG, which is transferred from the gut to the blood within the first 36 hours of life and provides a passive humoral immune level of protection for the piglet.
After this 36-hour window, the level of IgG decreases and the level of IgA rises, which is important in protecting the intestinal mucosal surface against infections (Markowsca-Daniel et al., 2010). Increasing IgG in colostrum is particularly important for first parity sows, as the level of IgG in the colostrum is 55% lower than that from multiparous sows (Cabrera et al., 2012.). This potentially drives the increased pre-weaning (14.4 vs. 11.2%) and post-weaning (5.6 vs 2.7%) mortality of gilt progeny compared with sow progeny (Smits, 2011).
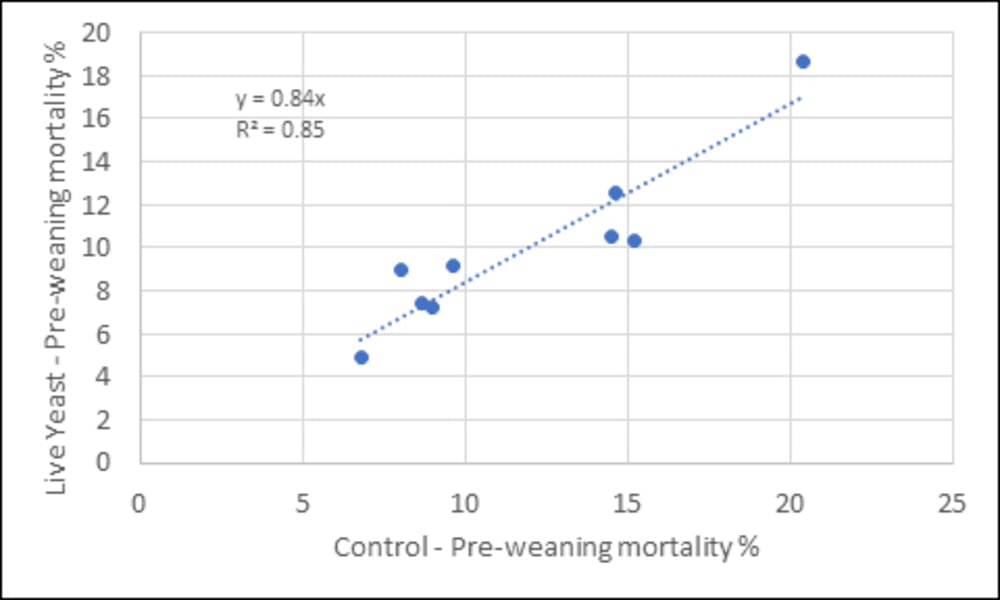
In a set of studies (AB Vista), the pre-weaning mortality of piglets was measured from sows that were fed with or without live yeast through lactation and compared (Figure 2). The results show that, on average, there is a 16% reduction in pre-weaning mortality (i.e. 10% to 8.4%) with the addition of a live yeast. On this basis, sows with 15 pigs born alive and a 10% pre-weaning mortality would be estimated to have an extra 0.24 pigs weaned per litter or an extra 0.6 piglets extra per sow per year.
Figure 2. Relationship in pre-weaning piglet mortality (%) between sows fed a control diet of that diet supplemented with 0.5 kg/t (> 24 d weaning) or 1 kg/t live yeast (< 24 d weaning)
In conclusion, with a greater number of piglets per litter, applications such as the use of a live yeast in gestation and lactation sow diets can be used to help mitigate the higher percentage of lower piglet birth weights and to improve the number of pigs weaned.
Latest news
Stay ahead with the latest news, ideas and events.
Online Feed Fibre Calculator
Calculate the percentage of dietary fibre in your feed
Our calculator is designed for nutritionists and uses averages of global raw materials to calculate the dietary fibre content (plus other more in-depth fibre parameters) of finished animal feed. These parameters are available within AB Vista’s Dietary Fibre analysis service (part of our NIR service).
Sign up for AB Vista news
A regular summary of our key stories sent straight to your inbox.
SUBSCRIBE© AB Vista. All rights reserved 2025
Website T&Cs Privacy & Cookie Policy Terms & Conditions of Sale University IDC policy Speak Up Policy